Figure 1: Air-cooled chiller located on a roof top.
A HVAC/R coating is used to protect the aluminium fins of a heat exchanger coil from corrosion whilst operating in an aggressive service environment. Despite the protection, filiform corrosion may still propagate at the aluminium-coating interface under certain environmental conditions, which can affect the operational efficiency of the HVAC/R asset. Filiform corrosion is an unidentified enemy to the vulnerable coated aluminium fins, as this type of corrosion is often seen, but not always understood – until now…
By Craig J R Thomas, HVAC/R Coating Specialist & Senior Managing Consultant – The Pipe-Sol Consultancy Ltd
Heat exchangers are designed and used to transfer heat from one medium to another in both heating and cooling processes, and one of their most widespread uses is for air conditioning and refrigeration. This type of heat exchanger is commonly referred to as a “coil”. Heating, ventilation, air conditioning and refrigeration (HVAC/R) systems employ various technologies to regulate temperature, humidity, and maintain air quality. HVAC/R systems provide heating and/or cooling for residential, commercial, industrial and institutional buildings. There are multiple types of HVAC/R unit (See Table 1), and the vulnerable coil is what links them all. The aluminium, copper, and galvanised iron metals that are used to manufacture the coil units are vulnerable to different types of corrosion.
It is very important that these critical HVAC/R assets are protected to maximise efficiency and extend life expectancy. High performance coatings are applied tothe aluminium/copper fins and copper tubes by original equipment manufacturers or specialist applicators. In fact, the whole unit can now be protected. One type of corrosion that is often seen, but not always identified and understood is Filiform corrosion (FFC).
The industry that is perhaps most affected by FFC is transportation, and in fact, it was described for the first time in 1944, whilst being observed on coated steel and then on high-resistance aluminium (alloys) used in aerospace¹. The HVAC industry and its aluminium fins are not however immune from attack from this type of corrosion.
Filiform corrosion – the basics
The aim of this article is to present a simple overview of filiform corrosion, including its incidence, appearance, and formation mechanism. Some of the implications of FFC relating to thin film organic coatings will also be discussed with specific reference to HVAC/R coatings.
Incidence and appearance of filiform corrosion
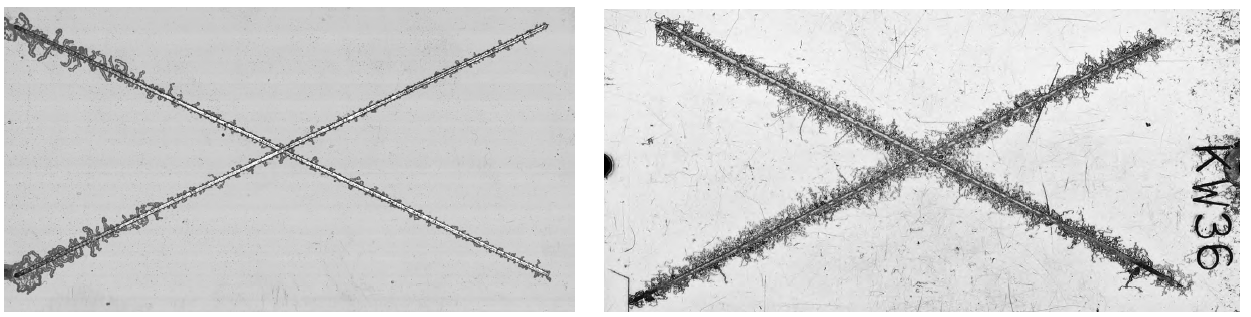
FFC can occur under thin film organic coatings that have a dry film thickness of less than 100 microns (4 mils) on both steel and aluminium (alloy) structures, although it is most commonly associated with aluminium substrates. The appearance of FFC is unique (see Figure 2) and bears a strong resemblance to fine filaments. These worm-like threads (100-500 microns (0.1-0.5 mm) wide and typically 1-10 mm long) emanate from one or more points (coating defects or damaged coating sites) in semi-random directions, which propagate at the metal-coating interface. The underlying metal, however, suffers only very superficial attack – generally of the order of tens of microns (maximum).
Mechanism of filiform corrosion
FFC typically initiates “when aggressive species, such as chloride ions, permeate through an organic coating, usually at a scratch, defect or weakness in the coating, and accumulate at the metal/coating interface”². The site of initiation tends to be an edge. Poor adhesion of a coating to an aluminium substrate or the presence of soluble salts under the coating at the time of application can also be responsible for FFC activity. Sodium chloride (sea salt) contains chloride ions and is classed as a soluble salt. FFC tends to be most prevalent in marine environments, although can occur in industrial locations. It can be accelerated by the presence of other soluble salts, which can enhance acidification at the corrosion site.
Very simply put, a “corrosion filament” comprises a head and tail, which form a mobile electrochemical (corrosion) cell on the surface of the aluminium. The head acts as the anode, and the tail is the cathode. The electrolyte is water, and the fourth element of the cell is aluminium. At the active head, aluminium is dissolved, and inert corrosion product is created in the form of aluminium hydroxide and pushed to the tail. Aluminium hydroxide is an amorphous powder, white in colour. If not recognised and left unchecked, corrosion filaments can propagate for months, or even years.
The factors that can accelerate the FFC process are usually increases in both temperature and relative humidity (RH). It has been postulated that the most rapid growth of corrosion filaments occurs at a RH of about 80%, allied to a temperature of around 40ºC (104ºF)³. The mechanism of filiform corrosion is shown schematically in Figure 3 in its most basic form.
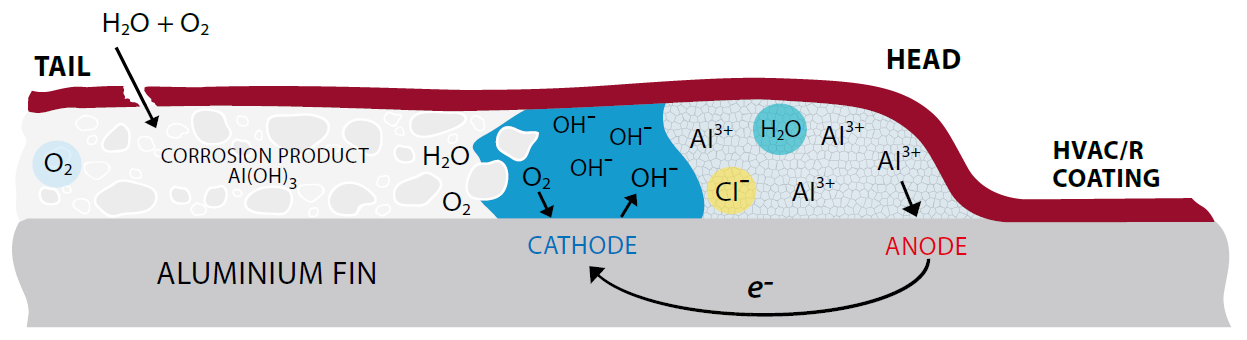
Formation of a corrosion filament – The techno-chemical version
The corrosion filaments comprise a mobile, electrolyte-filled head that itself comprises metal cations (Al3+) and aggressive anions (such as cl-), and a tail of dry, porous corrosion product.
At the leading edge of the filament head, anodic dissolution of aluminium occurs with the formation of aluminium cations (Al3+). transport of gaseous oxygen through the inert corrosion product of the tail continues to drive anodic metal dissolution at the leading edge of the filament head, whilst cathodic reduction of oxygen to form hydroxide anions (oH-) continues at the trailing edge of the head. And hence the formation of aluminium hydroxide (Al(oH)3) occurs. Aggressive anions (cl-) and water are conserved in the electrolyte in the filament head, thus enabling the corrosion filaments to propagate for lengthy periods of time (months, or even years) in the presence of a supply of oxygen.
Implications of filiform corrosion
FFC does not result in significant metal loss and is very much viewed by corrosion and coating specialists as a surface effect. However, its presence can be unsightly, and the expansive nature of the corrosion product formed result in bulging and delamination of the protective coating that has been applied. Selection of an appropriate surface preparation technique and a suitably compatible and robust material are therefore critical when considering protective coating systems for aluminium structures. In the case of HVAC/R units, where the fins are manufactured from aluminium, avoidance of filiform corrosion will be of paramount importance. The presence of corrosion product over a large area of the coil’s fin structure will have a significant effect on the HVAC/R asset’s performance.
When HVAC/R coils are coated, they become more resistant to corrosion and other forms of damage, which can help to improve their overall energy efficiency. This is because damaged or corroded coils are less efficient at transferring heat, which can lead to increased energy consumption and higher operating costs over time. By protecting the coils with a high-quality coating, businesses can reduce energy costs while also extending the lifespan of their equipment. Whilst it has been reported that FFC can occur with all paint and coating types¹, field experience suggests that certain coating types are inherently less susceptible to, and others demonstrate a greater propensity for FFC to occur. According to AMPP⁴, a high level of (wet) adhesion allied to a low water vapour transmission rate (WVTR) should minimise the propensity for FFC to initiate between the coating and the aluminium substrate at a (micro-)defect or site of damage. Standards form the foundation of testing and allow a better understanding of coating performance. ISO and ASTM have developed two similar, but not identical tests to determine the FFC resistance of organic coatings (on metal substrates).
Standard references identified in text
ASTM B117-19 – Standard Practice for Operating Salt Spray (Fog) Apparatus
ASTM D2803-09 (2020) – Standard Guide for Testing Filiform Corrosion Resistance of Organic Coatings on Metal
ISO 4623-2:2016 – Paints and varnishes – Determination of resistance to filiform corrosion. Part 2: Aluminium substrates
ISO 4628-10:2016 – Paints and varnishes — Evaluation of degradation of coatings — Designation of quantity and size of defects, and of intensity of uniform changes in appearance. Part 10: Assessment of degree of filiform corrosion
ISO 4623-2
The standard specifies a test, whereby scribe marks shall be cut through the coating applied to an aluminium panel, which are then exposed to a small amount of hydrochloric acid in the form of vapour. The panel is subsequently subjected to a temperature of 40ºC ± 2ºC (104ºF ± 4ºC) and a RH of 82% (± 5%) in a test cabinet. The test duration is not specified. FFC is only deemed to have occurred, if the typical pattern of worm-like threads or filaments is present. It is not possible to precisely rate FFC, so ISO calls for an assessment of filament length and frequency (ISO 4628-10) and suggests that photographs may be helpful in determining the extent of FFC.
Acronyms
AMPP – Association for Materials Protection and
Performance (formerly NACE International/SSPC )
ASTM – American Society for Testing and Materials
EHS – Environment, Health, and Safety
FFC – Filiform Corrosion
ISO – International Standards Organisation
RH – Relative Humidity
WVTR – Water Vapour Transmission Rate
VOC – Volatile Organic Compound
ASTM D2803
The standard guide calls for coated metal specimens to be scribed and placed in a corrosive atmosphere to initiate (filiform) corrosion. The specimens should then be exposed to controlled temperature and RH conditions “known to be conducive to filiform corrosion”. ASTM D2803 states that FFC usually occurs between 20-35°C (68-95°F) with a corresponding RH between 60-95%. The main differences between ASTM D2803 and ISO 4623-2 is that the ASTM standard guide uses salt spray (as per ASTM B117) to initiate corrosion as well as different temperature and RH ranges and specified test durations.
HVAC/R coil coatings
HVAC/R assets can be protected from highly corrosive and perhaps chemically aggressive service environments through the application of a high performance HVAC/R coating to the coil unit.
When considering the range of HVAC/R coatings on the market, although no specific coatings will be referenced in this article, it seems likely that chemical-cured or baked coatings by virtue of their densely cross-linked structures would exhibit a significantly lower WVTR compared to quick-(air)-dry coatings, and thus be more robust with respect to moisture permeation and hence resistant to FFC. It could also be anticipated that these chemically cross-linked products would afford superior “wet” adhesion compared to those air-drying materials, thus rendering them less susceptible to deformation or delamination in the event of FFC being initiated.
There is a growing trend in the industry to balance absolute performance from a coating solution with a solution that provides a more environmentally friendly impact on our world and is inherently safer to apply by coating technicians (and dispose of) due to the water-based chemistry of the formulation. Water-based coatings contain fewer or zero VOCs (volatile organic compound) and other hazardous chemicals. Their application is not restricted by international EHS guidelines, although normal PPE should be worn.
Formulation know-how in the 21st Century has also allowed water-based (acrylic) coatings to be created with enhanced properties through the incorporation of special additives. One such additive can generate self-healing properties, so if a small area becomes compromised, the coating’s self-healing properties can activate and “repair” the damage. The additive reacts with moisture to form a protective layer over the site, thus helping to prevent the spread of corrosion. Advanced corrosion inhibitors can also be added as well as de-foaming agents to functionalise and optimise corrosion protection and application performance respectively. Water-based (acrylic) coil coatings as with all protective coatings do require some coating application know-how and are easy to apply using a variety of methods, including spray, flow coat (flooding) application, and even roller. This allows easy set-up in an OEM plant, at a coil coating applicator, or at site without major EHS issues.


Figures 4 and 5: Water-based acrylic HVAC/R coating applied to the coils of an air-cooled chiller (left) and rooftop packaged unit (right) in the Middle East. Courtesy of Aqua Aero Coatings BV (URL: https://www.aquaaero.net)
HVAC/R coating type and enhanced surface preparation
Whilst the selection of specific coating type may play a significant role in minimising the incidence of filiform corrosion, surface preparation could be argued is equally important. Thorough cleaning of the substrate, such as removal of soluble salts and any (stamping) oils, grease etc., should be considered an absolute minimum requirement. However, given the geometric complexity of aluminium-finned heat-exchanger units, an appropriate chemical pre-treatment or conversion process, prior to application of the coating, should also perhaps be given serious consideration to provide additional insurance against FFC, particularly where warm, humid, salt-laden service environments may be anticipated.
A silane pre-treatment can provide added protection against FFC. It works by creating a chemical bond between the aluminium substrate and the water-based (acrylic) coating, which enhances the coating’s adhesion and prevents water from penetrating the coating and reaching the aluminium surface.
Conclusion
Filiform corrosion does exist, and aluminium HVAC/R fins are not immune from attack, when protected by an organic coating. What is critical is that the basics are understood – when it occurs, what it looks like, how it forms/initiates, and the results once it has propagated. It can be identified. Its appearance is unique (fine filaments). And it can be tested for using ISO or ASTM standards. During the design process for the coil, it is fundamental that the right protective coating is specified. The fins may be susceptible to filiform corrosion when a high performance coating is applied to the aluminium substrate in conditions of high salt concentration, elevated temperature and raised humidity. This can result in the creation of unsightly white corrosion product, and this surface effect will affect HVAC coil operational efficiency. It is possible to mitigate the risk of occurrence of filiform corrosion through the selection of a HVAC/R coating system that offers superior wet adhesion with and barrier properties to prevent the permeation of moisture to the substrate. Correct and thorough surface preparation is also vital. Nowadays, one popular choice for this application is a water-based acrylic coil coating, where reliable, durable protection is sought coupled with strong eco and EHS credentials.
About the author
 Craig set up his own international consultancy business in 2016, focusing most recently on HVAC/R coatings. He has over 20 years’ experience in the field of protective coatings, and has worked with/ for 3M, Copon-E Wood, Jotun, Kansai Paint, and Heresite. He is a certified NACE Coating Inspector (Level 1) and an ASHRAE Associate Member. Craig is also a Brand Ambassador for JettyRobot – supplier of unique and patented robotic HVAC duct and pipe cleaning technology.
Craig set up his own international consultancy business in 2016, focusing most recently on HVAC/R coatings. He has over 20 years’ experience in the field of protective coatings, and has worked with/ for 3M, Copon-E Wood, Jotun, Kansai Paint, and Heresite. He is a certified NACE Coating Inspector (Level 1) and an ASHRAE Associate Member. Craig is also a Brand Ambassador for JettyRobot – supplier of unique and patented robotic HVAC duct and pipe cleaning technology.
Linkedin: https://www.linkedin.com/in/craig-thomas-26b17930/
References
1. VARGEL, C., “Corrosion of Aluminium”, Elsevier, 2004.
2. MCMURRAY, H. N. and WILLIAMS, G., “Under Film/Coating Corrosion”, Reference Module in Materials Science and Materials Engineering, Elsevier, 2016.
3. YASAKAU, K.A., ZHELUDK EVIC, M. L. and FERREIRA, M. G. S., “Role of intermetallics in corrosion of aluminium alloys. Smart corrosion protection” in Intermetallic Matrix Composites: Properties and Applications, Woodhead Publishing, an imprint of Elsevier, 2018.
4. “Corrosion Basics”, https://www.ampp.org/technicalresearch/impact/corrosion-basics, AMPP, Accessed: 17/02/2023.
About this Featured Story
This Featured Story was first published in Heat Exchanger World Magazine in April 2023. To read more Featured Stories and many other articles, subscribe to our print magazine.
Featured Stories are regularly shared with our Heat Exchanger World community. Join us and share your own Featured Story on Heat Exchanger World online and in print.


